When to use key cancer testing methods: choosing the most appropriate approach at the right time

When to use key cancer testing methods: choosing the most appropriate approach at the right time
Discover three commonly used cancer testing approaches — liquid biopsy, tissue biopsy, as well as germline genetic testing — and how each is used at different points in the patient journey.
Introduction
Liquid biopsy tests transform cancer care by enabling genetic testing from a simple blood draw. These next-generation sequencing (NGS) liquid biopsy assays can detect tumor DNA in blood to guide treatment decisions without invasive tissue biopsies[1][2]. In 2020, the FDA approved two comprehensive liquid biopsy tests — Foundation Medicine’s FoundationOne and Guardant Health’s Guardant360 — as companion diagnostics for cancer therapy selection[3].
These traditional liquid biopsy solutions have proven the value of blood-based NGS tests — with FDA-approved panels like FoundationOne Liquid CDx and Guardant360 CDx guiding therapy for lung, breast, and other cancers. However, their high cost and centralized testing model limit widespread adoption[4].
Avitia represents a next-generation approach — delivering similar precision oncology insights on-site at a dramatically lower cost, roughly 60% less expensive than current send-out tests[18]. For labs and oncology providers, partnering with Avitia means faster time to treatment, lower costs, and the ability to offer cutting-edge liquid biopsy tests in-house[21][25]. As liquid biopsy technology continues to advance into screening and monitoring applications, in-house models like Avitia’s are poised to drive better outcomes by making these innovations accessible to every patient, everywhere[22].
Avitia’s On-Site Liquid Biopsy Solution – Faster, Cost-Effective, and In-House
In solid tumor oncology, choosing the right test at the right moment can accelerate therapy, reduce costs, and improve outcomes. This guide compares tissue biopsy, liquid biopsy (ctDNA), and germline genetic testing, and explains when each delivers the most clinical value across diagnosis, treatment selection, recurrence monitoring, and survivorship. (BioMed Central)
1) At a Glance: Learn Cancer Test Types
Tissue biopsy – solid tumor sample
The diagnostic foundation in solid tumors. Tissue biopsy remains the cornerstone of cancer evaluation in solid tumors, providing essential information on tumor morphology, grade, and architecture, as well as enabling immunohistochemistry (e.g., PD‑L1) and tumor genomic profiling. It is required to establish a definitive diagnosis and to characterize tumor subtypes at presentation. Somatic molecular profiling from tissue also identifies guideline‑recommended genomic biomarkers for treatment selection. (PMCID JAMA arXiv)
However, tissue sampling has limitations. Procedures are invasive and may not always be feasible, particularly in anatomically challenging sites or in patients with comorbidities. Single‑site biopsies may not fully capture spatial heterogeneity within a tumor, and processing times can delay treatment decisions. (PMCID)
Despite these challenges, tissue biopsy remains essential at initial diagnosis and in scenarios where histologic transformation or disease progression is suspected, and it continues to serve as the reference standard against which other testing approaches are interpreted. (JAMA)
Liquid biopsy – circulating tumor DNA (ctDNA) in blood
A complementary, minimally invasive approach for molecular insight and monitoring. Liquid biopsy (ctDNA) enables the detection of tumor‑derived genetic alterations from a blood sample, providing insight into actionable mutations, clonal evolution, and mechanisms of acquired resistance. In solid tumors, it is particularly useful when tissue is unavailable, insufficient, or unsafe to obtain, and can support treatment selection in the appropriate clinical context. (PMCID BioMed Central JCI)
A key advantage of liquid biopsy is its ability to be repeated over time, enabling real‑time monitoring of disease dynamics, treatment response, and emerging resistance. Serial ctDNA assessment is well suited for longitudinal evaluation in advanced disease and can capture inter‑site tumor heterogeneity not reflected in a single biopsy. (PMCID JAMA)
However, sensitivity may be limited in early‑stage or low‑burden disease, where circulating tumor DNA levels are low or undetectable. Liquid biopsy also does not provide histologic information and does not replace tissue biopsy for initial diagnosis. Instead, it is best used as a complementary approach alongside tissue‑based testing. (PMCID BMC Cancer)
Germline genetic testing – blood or saliva
An assessment of inherited risk with implications for treatment and family care. Germline genetic testing evaluates inherited pathogenic variants associated with increased cancer susceptibility (e.g., BRCA1/2), providing information that can inform cancer risk assessment, prevention strategies, and tailored surveillance. In patients with solid tumors, germline findings may also influence treatment decisions in specific contexts (e.g., eligibility for targeted therapies such as PARP inhibitors). (Cancer.gov Jackson Laboratory)
Germline testing is typically considered based on tumor type, age at diagnosis, personal and family history, or the presence of suggestive tumor features. Results have implications beyond the individual patient, supporting cascade testing and risk assessment for family members. (Jackson Laboratory)
Unlike somatic tumor testing (from tissue or liquid biopsy), which characterizes acquired mutations in the tumor, germline testing identifies inherited variants present in all cells. The two approaches are complementary and are often used together to provide a more complete view of cancer risk and treatment considerations. (Cancer Med)
2) When to Test for Cancer: A Practical Framework
A. Initial diagnosis: suspected solid tumor
Tissue biopsy is the foundation at initial presentation, enabling definitive diagnosis, histologic classification, staging, and biomarker assessment (e.g., immunohistochemistry and tumor genomic profiling). It remains essential to establish tumor type and guide first‑line treatment decisions. (PMCID)
Liquid biopsy (ctDNA) can be used in parallel when tissue is limited, unsafe to obtain, or when rapid turnaround is critical. In this setting, it may help identify clinically relevant mutations to support earlier treatment selection, particularly in cancers where guideline‑recommended biomarkers are required before therapy initiation. (Liquid Biopsy FAQ)
Using tissue and liquid biopsy together at diagnosis can help mitigate sampling limitations and reduce delays, especially in advanced diseases where timely treatment decisions are needed. (JAMA)
B. On treatment: response assessment and resistance
During treatment, liquid biopsy enables minimally invasive, longitudinal monitoring of tumor‑derived DNA. Serial assessment of ctDNA can provide insight into treatment response, clonal evolution, and the emergence of resistance mutations—often capturing dynamic changes that a single tissue sample cannot. (PMCID)
However, liquid biopsy does not replace tissue in all scenarios. If ctDNA is not detected despite suspected progression, or if histologic transformation is a concern, repeat tissue biopsy is recommended to reassess tumor morphology and enable comprehensive analysis. (PMCID)
In practice, liquid biopsy supports real‑time molecular monitoring, while tissue biopsy remains important when phenotypic changes or diagnostic confirmation are required. (PMCID)
C. Post‑treatment monitoring: minimal residual disease and recurrence
After curative‑intent therapy, liquid biopsy is increasingly used to detect minimal residual disease (MRD) and to identify recurrence earlier than standard imaging in some solid tumors. Detection of ctDNA following treatment is associated with a higher risk of relapse and may inform risk‑adapted surveillance strategies. (PMCID)
Emerging evidence also supports the use of ctDNA to guide adjuvant therapy decisions in select settings, helping to refine treatment intensity. However, clinical adoption varies by tumor type, and results should be interpreted within the context of established guidelines and other clinical findings. (PMCID)
D. Inherited risk assessment and broader care considerations
Germline genetic testing should be considered when personal or family history, tumor type, or clinical features suggest an inherited cancer predisposition (e.g., early‑onset disease, multiple primary cancers, or characteristic tumor profiles). (Jackson Laboratory)
Germline findings inform cancer risk assessment, screening, and prevention strategies, and may also influence treatment decisions in specific contexts. Results have implications beyond the individual patient, enabling risk assessment and cascade testing for family members. (Jackson Laboratory)
Importantly, germline and somatic testing are complementary. Tumor‑based testing (from tissue or liquid biopsy) identifies acquired mutations that guide therapy, while germline testing distinguishes inherited variants and provides a broader view of cancer risk. (NIH)
3. Practical Decision Table for Solid Tumor Testing
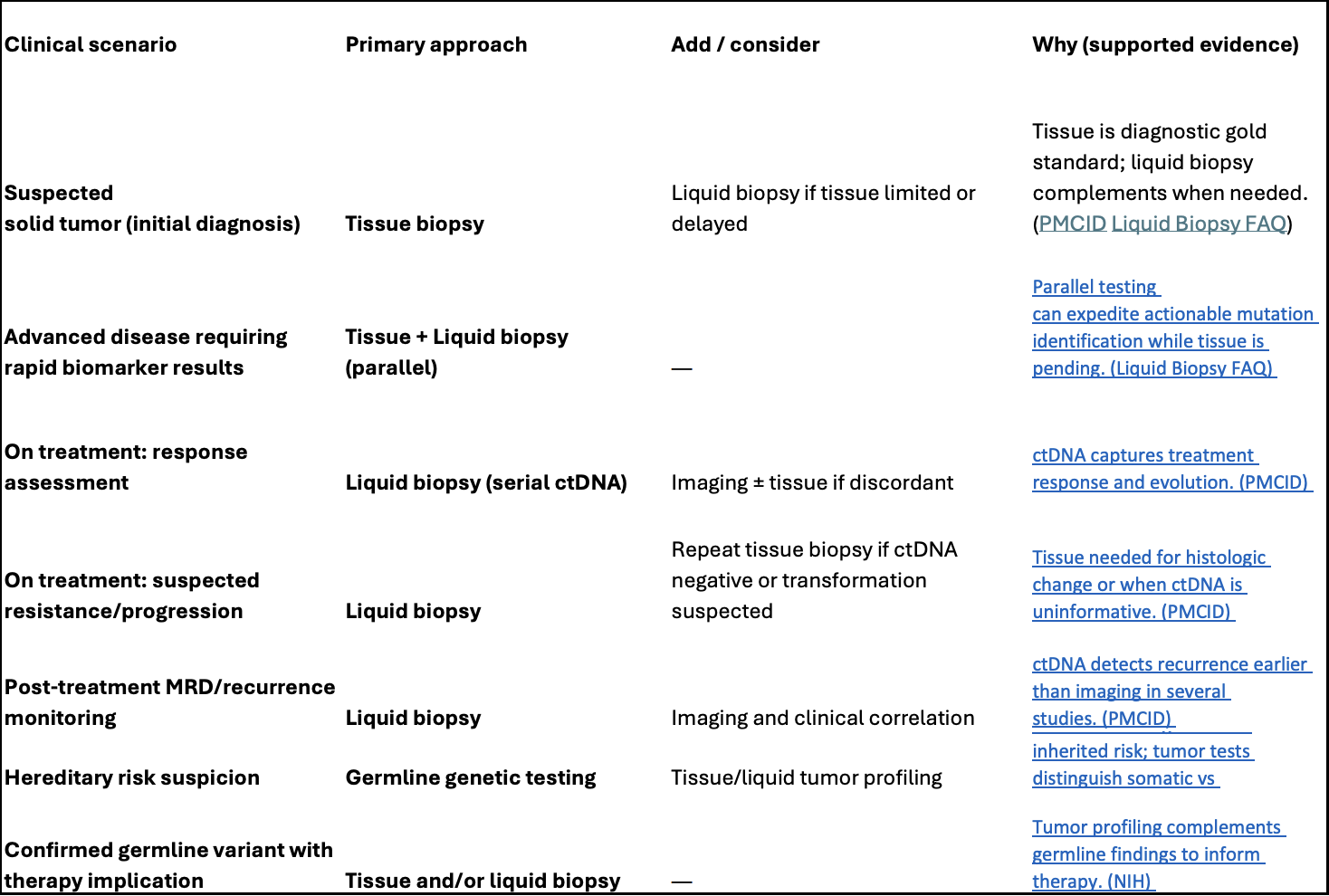
In practice, these approaches are not interchangeable but complementary: tissue biopsy establishes diagnosis, liquid biopsy enables dynamic molecular monitoring, and germline testing provides inherited risk context. Using them together — at the right time — supports more informed and timely decision-making in solid tumor care.
4) What leading centers emphasize
- MSK: Liquid biopsies are already useful for treatment monitoring; robust programs continue in early detection and minimal residual disease research. (Memorial Sloan Kettering Cancer Center)
- ESMO (2024): Structured, clinician-friendly genomic reports improve the utility of tumor profiling—whether from tissue or liquid. (annalsofoncology.org PMC)
5) Avitia’s role in access, speed, and equity
Avitia’s distributed, AI-powered platform enables hospital and regional labs to perform NGS liquid biopsy on-site — reducing shipping delays and costs while making repeat testing more practical and accelerating clinical decision-making. Keeping testing within the country of care also gives clinicians access to more representative, locally generated genomic data — offering better insight into how cancer presents across different populations and subpopulations than centralized lab models. These insights support more informed treatment decisions and earlier preventive action, with particular impact in community settings where most patients receive care. By keeping patient data local, Avitia further enables region-specific research, supports the development of local clinical trials, and generates insights tailored to each population’s unique needs—advancing data sovereignty while improving patient outcomes. (Avitia Press Release,Avitia Liquid Biopsy Blog,Avitia Health Equity Blog)
6) Clinician checklist
- At first presentation
Order tissue biopsy for definitive diagnosis, histology, immunohistochemistry, and tumor genomic profiling. Consider adding liquid biopsy (ctDNA) in parallel if tissue is limited, unsafe to obtain, or if rapid molecular insights are needed.
- Before major treatment decisions
Ensure current tumor genomic information is available. Use tissue and/or liquid biopsy as appropriate—liquid biopsy may provide faster results in some settings and can be repeated to capture evolving mutations.
- If progression is suspected
Use liquid biopsy to assess for resistance mutations and clonal evolution. Escalate to repeat tissue biopsy if ctDNA is not detected, results are inconclusive, or histologic transformation is suspected.
- After curative-intent therapy
Consider ctDNA testing for minimal residual disease (MRD) and early detection of recurrence in appropriate solid tumors. Interpret results in the context of imaging, clinical findings, and current guidelines.
- If hereditary risk is suspected
Order germline genetic testing with appropriate counseling. Use results to inform risk assessment, surveillance strategies, potential treatment implications, and cascade testing for family members.
7) Conclusion
No single testing approach answers every clinical question in solid tumor care. Tissue biopsy, liquid biopsy (ctDNA), and germline genetic testing each provide distinct and complementary insights that support decision-making at different points in the patient journey.
Tissue biopsy remains the diagnostic foundation, establishing tumor type, histology, and key biomarkers. Liquid biopsy adds a minimally invasive way to assess tumor genomics over time, offering insight into treatment response, clonal evolution, and emerging resistance. Germline genetic testing provides a broader perspective on inherited cancer risk, with implications for prevention, treatment selection in specific contexts, and family care.
Used together — and applied at the right time — these approaches enable a more complete understanding of both the tumor and the patient. As evidence continues to evolve, integrating tissue, liquid, and germline testing into clinical workflows can support more informed, timely, and individualized care in solid tumors.
References:
- NCI. Genetic Testing for Inherited Cancer Risk — Fact Sheet (updated 2024). Cancer.gov
- American Cancer Society. Understanding Genetic Testing for Cancer Risk (2024). American Cancer Society
- Tie J, et al. ctDNA-Guided Adjuvant Therapy in Stage II Colon Cancer (NEJM, 2022). nejm.org+1
- Parikh AR, et al. Liquid versus tissue biopsy for detecting acquired resistance and tumor heterogeneity (Nat Med, 2019). PMC
- van de Haar J, et al. ESMO Recommendations on clinical reporting of genomic tumor profiling (Ann Oncol, 2024). annalsofoncology.org+1
- MSK. Liquid Biopsies and Other Blood Tests for Cancer (patient education, 2025). Memorial Sloan Kettering Cancer Center
- Avitia. Liquid Biopsy Tests — A Cost-Effective Shift in Cancer Diagnostics; Platform/Press (on-site NGS; access & equity). avitia.bio+1
- Caputo V, Ciardiello F, Corte CMD, Martini G, Troiani T, Napolitano S. Diagnostic value of liquid biopsy in the era of precision medicine: 10 years of clinical evidence in cancer. Explor Target Antitumor Ther. 2023;4(1):102-138. doi: 10.37349/etat.2023.00125. Epub 2023 Feb 28. PMID: 36937316; PMCID: PMC10017193.
- Iams WT, Mackay M, Ben-Shachar R, Drews J, Manghnani K, Hockenberry AJ, Cristofanilli M, Nimeiri H, Guinney J, Benson AB 3rd. Concurrent Tissue and Circulating Tumor DNA Molecular Profiling to Detect Guideline-Based Targeted Mutations in a Multicancer Cohort. JAMA Netw Open. 2024 Jan 2;7(1):e2351700. doi: 10.1001/jamanetworkopen.2023.51700. PMID: 38252441; PMCID: PMC10804266.
- Jahani, M.M., Mashayekhi, P., Omrani, M.D. et al. Efficacy of liquid biopsy for genetic mutations determination in non-small cell lung cancer: a systematic review on literatures. BMC Cancer 25, 433 (2025). https://doi.org/10.1186/s12885-025-13786-w
- The Jackson Laboratory, Indications for Genetic Testing for Inherited Cancer Risk After Biomarker Testing
- DeBortoli E, McGahan E, Yanes T, Berkman J, Aoude LG, Smit AK, Gokoolparsadh A, Hermes A, Newett L, Bourke M, Hanson S, Hughes H, Hofmann O, Goranitis I, McWhirter R, Milch V, Steinberg J, McInerney-Leo A. Utility of Germline, Somatic and ctDNA Testing in Adults With Cancer. Cancer Med. 2025 Aug;14(15):e71080. doi: 10.1002/cam4.71080. PMID: 40747594; PMCID: PMC12314551.
- The Jackson Laboratory, Liquid Biopsy FAQ
Gardez une longueur d'avance sur le cancer dès aujourd'hui

