Fast, Fair, Cancer Care: Why Liquid Biopsy Must Be Publicly Reimbursed — and Why 2026 Is the Year to Act
_Campaign_Post_END%20(1).png)
Fast, Fair, Cancer Care: Why Liquid Biopsy Must Be Publicly Reimbursed — and Why 2026 Is the Year to Act
Cancer doesn’t discriminate. It doesn’t care where you live, what you earn, or which health system you were born into. However, access to advanced diagnostics still does.
Today, Avitia is launching Fast, Fair, Cancer Care — a global initiative beginning in Canada — with one clear call to action:
Improve access to advanced cancer testing by making liquid biopsy publicly reimbursed to help guide care.
The science is proven, the technology exists, and the data on cost savings is unambiguous. What’s missing is the systemic ability to act.
Spread the word:
Fast, Fair, Cancer Care SocialToolkit
What Is Liquid Biopsy — and Why Does It Change Everything?
Liquid biopsy is one of the most significant advances in cancer diagnostics in a generation. Instead of cutting into tissue to find cancer mutations — an invasive, often delayed, and logistically complex procedure — a liquid biopsy begins with something as routine as a blood draw.[1][2]
That blood sample is then analyzed using genomic technologies — including next-generation sequencing (NGS) — to detect circulating DNA (ctDNA). The result is a detailed molecular profile of a patient’s cancer — the specific mutations driving it, the therapies most likely to work, and the early signals of whether a cancer is returning.[1][3]
For patients with advanced diseases like lung, breast, or colorectal cancer, this information is not a nice-to-have. It is the difference between receiving the right targeted therapy or spending weeks on treatments that won’t work. It is the difference between detecting a recurrence early or discovering it after it has progressed. It is, in the most literal sense, the difference that can save lives.[4][5]
.png)
The Problem: Advanced Diagnostics Aren’t Reaching Patients
Despite clear clinical evidence, liquid biopsy remains inaccessible to the majority of cancer patients across Canada and much of the world. The barriers are systemic, not scientific.[6]
- Reimbursement gaps leave patients behind. Without public funding, liquid biopsy testing is available primarily to those who can access it through private payers or research programs — creating a two-tier system for one of the most consequential decisions in cancer care: what treatment to receive.[6][7]
- Geography compounds inequity. In Canada — the second-largest country in the world by area — the standard model for advanced genomic testing requires shipping blood samples to centralized urban labs. For patients in rural and remote communities, this adds logistical complexities, days — if not weeks — of delays, and the very real risk that treatment decisions are made without complete genomic information.[1][8]
- Delays in biomarker testing have direct consequences. Published research shows that delays in tissue biopsy and molecular testing have been associated with 17% of patients dying or becoming unsuitable for treatment — and 49.7% of patients not receiving targeted therapies due to delayed biomarker results. Every day between diagnosis and the right treatment matters.[9][10]
- Most Canadian cancer patients are not being tested. Despite national and international guidelines recommending biomarker testing for lung, breast, and colorectal cancers, a recent real-world analysis found that fewer than 30% of Canadian cancer patients receive molecular testing.[11][12] In the pan-Canadian ACTT program, 49% of patients enrolled had received no previous molecular testing at all.[1]
The conclusion is stark: most Canadian cancer patients are making treatment decisions without the genomic information that could support their oncologists and guide them to better outcomes.
What the Evidence Says: Pan-Canadian Liquid Biopsy Study
The case for public reimbursement is not theoretical. It is built on three years of real-world evidence from across Canada.
Between 2020 and 2023, Avitia launched Project ACTT (Access to Cancer Testing & Treatment), a pan-Canadian ctDNA genomic testing program. The results, published in Current Oncology (Lapuk et al., 2026), represent a large, real-world evidence study of what liquid biopsy can achieve at a national scale.[1]
The scale: 4,229 patients. 150+ institutions. 12 provinces.
Over three years, 4,229 patients with advanced solid tumors submitted samples for liquid biopsy testing. Reports were successfully delivered for 97% of participants, with a median turnaround time of 8 days from sample receipt to results.[1] Critically, 82% of all registered oncologists in Canada — working across more than 150 institutions — participated in the program. Eleven percent of samples came from outside major urban centers, including remote communities across British Columbia, Northern Ontario, Atlantic Canada, and beyond.[1]
This was not a controlled trial in ideal conditions. This was the Canadian healthcare system as it operates — and liquid biopsy worked.
The clinical impact: actionable results for the patients who needed them most
Across the cohort of lung, breast, and colorectal cancer patients:[1]
- ctDNA mutations were detected in more than 50% of cases
- Clinical trial matches were identified for 75% of participants with an identified mutation
Among patients with no prior molecular testing, liquid biopsy identified actionable mutations in hundreds of patients who might otherwise have received suboptimal treatment
The economic case: liquid biopsy saves the system money
Perhaps the most important finding for health system decision-makers: adding liquid biopsy to the standard tissue biopsy pathway does not cost the system more. It can save money.[1][13]
A health economics model developed in collaboration with the Institute of Health Economics (Edmonton, AB), focused on advanced non-small cell lung cancer (NSCLC) patients in Ontario, found:[1]
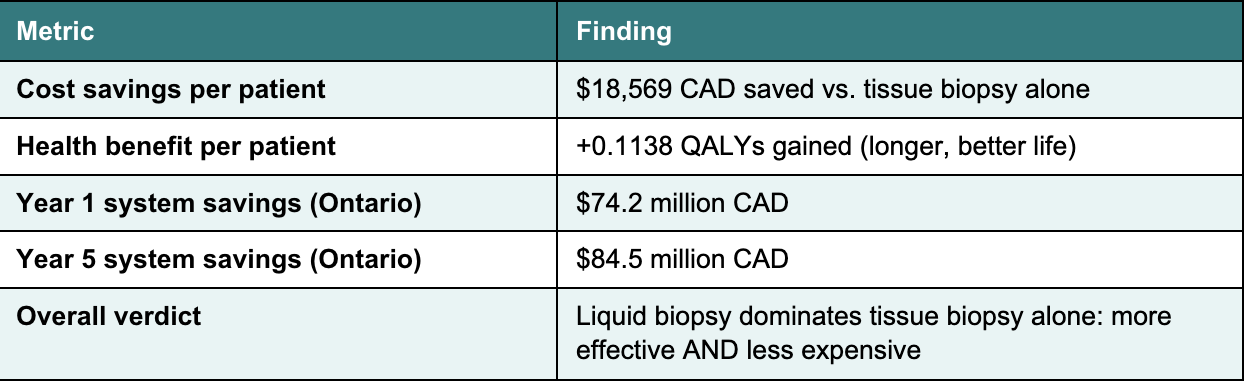
Source: Lapuk et al., Current Oncology, 2026[1]; Institute of Health Economics modeling, Ontario NSCLC cohort.
The majority of cost savings come from more precise treatment selection: getting patients onto the right targeted therapy faster means fewer costly trial-and-error chemotherapy combinations, fewer adverse events, and reduced downstream healthcare utilization.[1][13] The test pays for itself many times over.
This is what value-based cancer care looks like in practice.
What Fast, Fair, Cancer Care Means for Patients in Rural and Underserved Communities
One of the most important findings from Project ACTT was the reach into communities that centralized testing models have historically left behind. Eleven percent of the program’s samples came from outside major urban centers.[1]
This matters because the promise of precision oncology has largely been an urban one. If you live near a major cancer center, you are far more likely to receive comprehensive biomarker testing. If you don’t, you are far more likely to be treated without it.[6][8]
Avitia’s in-house platform — bringing NGS liquid biopsy technology directly to local labs rather than shipping samples to distant facilities — is designed to close this gap. When a community hospital can run its own liquid biopsy test on site, it no longer depends on sample transit times, centralized lab queues, or the logistical complexity of long-distance sample shipping. The result is faster results, lower costs, and care that reaches patients where they actually live.[1][13]
This is more than efficiency. It is about equity.
Every Cancer Month Is a Reminder. Fast, Fair, Cancer Care Is the Support.
Every month of the year, the world recognizes different cancers. Pink ribbons for breast cancer. White for lung. Dark blue for colorectal. These moments of awareness are meaningful — they reduce stigma, encourage screening, and build community for those living with cancer.
Our goal is to support oncologists with powerful molecular information they need to provide patients with the best possible care.[6][7]
Fast, Fair, Cancer Care says: we know what works. We have the evidence. We have the technology. The question now is whether our health systems will fund it — not as a research program, not as a pilot, but as standard of care.
Our call to action:
- To health system leaders: The economic modeling is clear. Reimbursing liquid biopsy saves money and saves lives. Canada’s Drug Agency (CDA) has already identified this technology as having significant potential impact on Canada’s healthcare system.[14] Act on the evidence.
- To oncologists and clinicians: 82% of Canadian oncologists who participated in Project ACTT ordered liquid biopsy testing.[1] That adoption rate speaks to clinical confidence. Advocate for your patients’ access to this tool as standard practice.
- To patients and advocates: You have the right to the most accurate diagnostic information available to guide your care. Ask your care team about liquid biopsy. Demand it be covered.[7]
- To policymakers: Single-payer and hybrid health systems are uniquely positioned to implement liquid biopsy equitably — reaching every patient, not just those who can pay. Canada can show the world what fast and fair cancer care looks like.[6]
Join Fast, Fair, Cancer Care
Avitia will be sharing educational resources throughout April and beyond for anyone interested in value-based healthcare, liquid biopsy reimbursement, and how diagnostics shape both clinical and system-wide outcomes.
Amplify the message. Post about Fast, Fair, Cancer Care this April — and every month a cancer is recognized.
Stand with us — and with patients everywhere. Access the campaign messaging and social toolkit here:
Fast, Fair, Cancer Care SocialToolkit
The data is in. The economics are clear. The patients are waiting.
It’s time for fast, fair cancer care — for everyone.
Together in Fast, Fair, Cancer Care
Sources:
- Lapuk A, et al. – Lessons from a National Liquid Biopsy Program (Project ACTT), Current Oncology, 2026 [1]
- Rolfo C, et al. – Liquid Biopsy for Advanced NSCLC: A Consensus Statement, Journal of Thoracic Oncology, 2021 [2]
- Hench IB, et al. – Liquid Biopsy in Clinical Management of Breast, Lung, and Colorectal Cancer, Frontiers in Medicine, 2018 [3]
- Breadner D, et al. – Implementation of Liquid Biopsy in NSCLC: An Ontario Perspective, Current Oncology, 2024 [4]
- Juergens RA, et al. – Demonstrating the Value of Liquid Biopsy for Lung Cancer in a Public Health Care System, Journal of Clinical Oncology, 2020 [5]
- Sheffield BS, et al. – Toward Timely and Equitable Advanced Biomarker Testing for Patients with Metastatic Cancer in Canada, Current Oncology, 2025 [6]
- Colorectal Cancer Canada – Patient & Caregivers’ Perspective on Biomarker Testing Across Canada, Survey Results, 2021 [7]
- Husereau D, et al. – Progress toward Health System Readiness for Genome-Based Testing in Canada, Current Oncology, 2023 [8]
- Elkrief A, et al. – Therapeutic Landscape of Metastatic NSCLC in Canada in 2020, Current Oncology, 2020 [9]
- Sadik H, et al. – Impact of Clinical Practice Gaps on the Implementation of Personalized Medicine in Advanced NSCLC, JCO Precision Oncology, 2022 [10]
- Lee TH, et al. – Real-world analysis of biomarker testing timing in metastatic colorectal cancer, Journal of Clinical Oncology, 2025 [11]
- Husereau D, et al. – Progress toward Health System Readiness for Genome-Based Testing in Canada, Current Oncology, 2023 [12]
- Patel YP, et al. – Health and Budget Impact of Liquid-Biopsy-Based CGP Testing in Tissue-Limited Advanced NSCLC, Current Oncology, 2021 [13]
- Basharat S, Farah K – An overview of comprehensive genomic profiling technologies to inform cancer care, Canada’s Drug Agency (CDA), 2022 [14]
Fast, Fair, Cancer Care is a global advocacy initiative launched by Avitia in 2026, beginning in Canada, calling for the public reimbursement of liquid biopsy testing as part of standard cancer treatment selection pathways. Learn more at www.avitia.bio
Stay Ahead of Cancer Today

